“Evidence of widespread metabolite abnormalities in Myalgic encephalomyelitis/chronic fatigue syndrome: assessment with whole-brain magnetic resonance spectroscopy”
PI: Jarred Younger, PhD
University of Alabama at Birmingham
- Home
- >
- Jarred Younger, PhD
JARRED YOUNGER
“Evidence of widespread metabolite abnormalities in Myalgic encephalomyelitis/chronic fatigue syndrome: assessment with whole-brain magnetic resonance spectroscopy”
PI: Jarred Younger, PhD
University of Alabama at Birmingham

Researchers from the Neuroinflammation, Pain and Fatigue Laboratory, led by Jarred Younger (PhD) at the University of Alabama at Birmingham examined inflammatory processes at work in the brains of people with ME/CFS. In collaboration with University of Miami Miller School of Medicine radiology experts, the researchers employed novel use of a neuroimaging tool, magnetic resonance spectroscopy (MRS). Dr. Younger’s pilot study provides compelling evidence that low-level neuroinflammation – present across a range of brain areas – is a factor in ME/CFS. The results corroborate previous observations of brain abnormalities, including elevated lactate, and extend findings from MRS studies that were limited to specific regions of the brain.
Major Ramsay goal fulfilled:
✓ Received an R01 grant from NIH. Dr. Jarred Younger’s pilot data was used in a proposal to the National Institutes of Health to run an expanded study that was funded in 2019.
✓ Added to the cumulative, scientific knowledge. The study “Evidence of widespread metabolite abnormalities in Myalgic encephalomyelitis/chronic fatigue syndrome: assessment with whole-brain magnetic resonance spectroscopy” was published in Brain Imaging and Behavior. Read it here.
Publication in Brain Imaging & Behavior
>>> What you need to know
- Jarred Younger, lead author Christina Mueller (M.S.), and co-authors completed a neuroimaging study of a female cohort of 15 individuals with ME/CFS (who were carefully screened and met a modified Fukuda case definition) and 15 age-matched healthy controls using magnetic resonance spectroscopy (MRS).
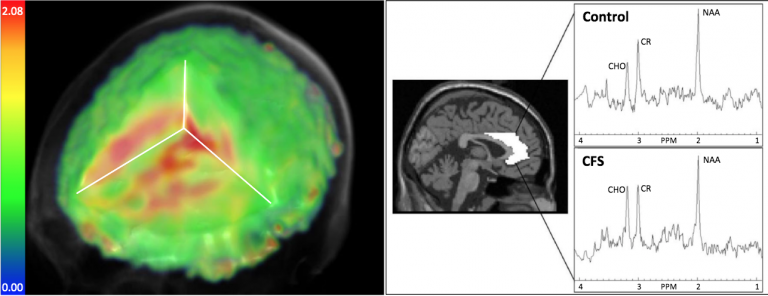
- MRS, a type of MRI scan, provides a non-invasive method for evaluating the types and quantities of chemicals in the brain using 3D images and can give a readout of metabolic changes.
- The researchers found that “metabolite and temperature abnormalities were distributed across large portions of the brain” in ME/CFS participants, as compared to controls.
- The most significant finding was elevation of choline in the anterior cingulate (ACC) area of the brain on the left side. Increases in choline are associated with immune cell activation and the authors note that previous research indicates a critical role for the ACC region in cytokine-induced fatigue.
- Lactate (a byproduct of glycolysis in an oxygen-limited environment) was found to be increased in a number of brain areas, consistent with brain inflammation and an energy deficit at the cellular level.
- Higher average temperatures were observed in five brain areas; the researchers included assessments that showed this finding wasn’t attributable to differences in blood flow or whole-body temperature. Inflammation requires more metabolic expenditures and three of the five areas also measured increased lactate, suggesting increased metabolism that could be related to neuroinflammation.
- The authors acknowledge a few limitations of the study, including the small sample size, but these preliminary results support a hypothesis of neuroinflammation in ME/CFS and provide a benchmark for replication using larger study groups.
Learn more about the Ramsay Grants

Meet the Researchers
Learn more
Apply for a Grant
Learn more